Recent news in Food & Feed Analysis
- Home
- /
- Microorganisms in cosmetics: Can...
Microorganisms in cosmetics: Can your make-up make you ill?

Microbiological contamination by bacteria, molds or viruses may not only occur in food. Pharmaceutical and cosmetic products may be contaminated, too. Microorganisms spoil the product and may be hazardous for consumer health.
Cosmetics provide an optimal medium for microbial contaminants: With proteins, sugar, vitamins, oils and water, they contain everything microorganisms need to grow. The neutral pH-value and the storage in a warm and humid bathroom contribute to an ideal climate for bacteria and molds. Via hand and mouth, a lot of microorganisms get into the product. Sometimes the product is already contaminated during the production process, for example from contaminated raw materials. Most of the microorganisms are harmless to the consumer. However, microbial contaminants can cause dangerous infections in infants and elderly or sick people. In addition, some molds and bacteria produce toxins which can cause allergic reactions and skin irritations.
In order to minimize the microbial contamination, the IKW recommends to take the following actions in connection with a Microbiological Quality Management:
- Use of the principles of good manufacturing practice
- Application and verification of limit values
- Use of preservatives
Preservatives in cosmetics: Important or harmful?
Preservatives are essential in the production of cosmetics, because they kill or inhibit the growth of microorganisms. Without preservatives, most aqueous cosmetic products would have a shelf life of only 2 weeks. Only a few cosmetic products are stable without added preservatives. These include products with essential oils or a high level of alcohol as well as products that contain little or no water.
Preservatives are therefore very important to keep cosmetics from spoiling and to inhibit the growth of potentially dangerous microorganisms. However, not all substances that can be used as a preservative are harmless to the consumer. Parabens for example have a hormonal effect; benzoic acid can cause pseudoallergic reactions. The German Cosmetics Regulation lists all preservatives allowed in cosmetic products.
In addition, there are a couple of natural materials that contain substances that are said to have an antimicrobial effect. These include for example propolis, cinnamon bark, clove oil, allspice oil, nutmeg oil, ylang ylang, rose oil and thyme. However, these substances have not been adequately researched for use as a preservative in the cosmetics industry.
Methods of microbiological analysis of cosmetic products
At the moment, there are no legally binding limit values for germ contents in cosmetics. According to a recommendation of the Scientific Committee on Consumer Safety of the European Commission, cosmetics should contain no more than 100 or 1.000 colony-forming units per gram, depending on the type of product. Certain pathogens such as staphylococcus or E. coli are not allowed to be detectable at all.
To ensure that these limit values are adhered to, you can easily analyze the product during the production process. An appropriate instrument for this purpose is a culture medium plate, on which the sample is placed. After an incubation period of 24 – 48 hours, the bacterial count can be measured.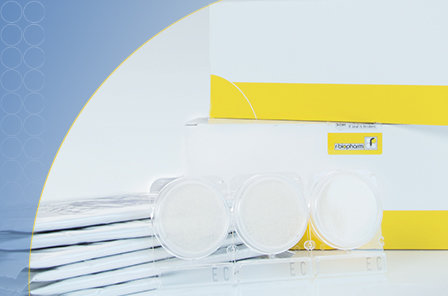 With our new Compact Dry for Cosmetics series, such test kits for the microbiological analysis of creams, lotions, lipsticks and other cosmetic products are now available at R-Biopharm. With these tests, you can measure the total bacteria count as well as the amount of E. coli, coliforms and staphylococcus aureus in cosmetics.
With our new Compact Dry for Cosmetics series, such test kits for the microbiological analysis of creams, lotions, lipsticks and other cosmetic products are now available at R-Biopharm. With these tests, you can measure the total bacteria count as well as the amount of E. coli, coliforms and staphylococcus aureus in cosmetics.
Our products for the microbiological analysis of cosmetics:
- Compact Dry TC (total aerobic count)
- Compact Dry EC (coliform bacteria and E. coli)
- Compact Dry CF (coliform bacteria)
- Compact Dry X-SA (staphylococcus aureus)