Constituents
Precise determination of acid, sugar and alcohol content
Constituents
Enzymatic tests for food analysis
Enzymatic tests are essential analytical tools used for the determination of sugars, acids, alcohols, and other food components in a wide range of products, including fruit juices, wine, beer, dairy products, eggs, and meat. These tests are highly specific and effective even in complex matrices, thanks to the use of high-quality enzymes that ensure precise and accurate measurements. The results of these enzymatic assays are measured using a spectrophotometer, and the process can be automated for efficiency.
Validation and Approval by International Organizations
Numerous enzymatic methods have been validated or approved by leading international organizations, ensuring their reliability and standardization in food analysis. The most important of these organizations include:
- AOAC (American Association of Analytical Chemists)
- CEN (European Committee for Standardization)
- IDF (International Dairy Federation)
- IFU (International Federation of Fruit Juice Producers)
- ISO (International Standardisation Organisation)
- OIV (International Organization of Wine)
Comprehensive test systems for enzymatic analyses
We offer a diverse range of test systems for enzymatic analyses, including the internationally recognized reference method “Yellow Line” by Roche. Our product lines also include the Enzytec™ series—“Generic,” “Color,” “Standards,” and “Liquid”—as well as test kits designed for the convenient on-site biochemistry analyzers RIDA®CUBE SCAN and Pictus 500.
Importance of acid determination
Determining the levels of organic acids such as citric acid, lactic acid, and acetic acid is crucial in the production of beverages like wine and beer. Some acids can spoil the product, others are used as additives, and still others serve as important quality indicators. Understanding the role of these acids in food production is essential for maintaining product quality and safety. Click on one of the following acids to learn more about its significance in food production.
Acetic acid is an important metabolic analyte, the end product of fermentation processes and the oxidation product of acetaldehyde and ethanol. Determination of acetic acid is particularly important in the wine industry: As a sensory compound, it adds flavor and complexity in small amounts, but spoils the wine at high concentrations. Acetic acid is the main component of the “volatile acids” in wine and is measured throughout the entire vinification process. Vinegar is roughly 3 – 9 % acetic acid by volume and gives the monetary value of this product. Acetic acid is also used in food production as a preservative (E260) and as taste-improver. Therefore, it is tested in a wide range of food products such as beer, bread and bakery goods, baking agents and sourdough, dairy, fish, fruit and vegetable products, meat, ketchup, mayonnaise, mixed pickles, salts and spices, sauces and dressings, soy sauce, tea and vinegar as well as in animal feed and pharmaceuticals.
L-ascorbic acid (vitamin C) is a naturally occurring organic compound with antioxidant properties and is found ubiquitously in fruit and vegetables. Its quantitative determination is especially important in the production of wine, beer, milk, soft drinks and fruit juices, where it can be a quality indicator. Given the essential role in human diet, L-ascorbic acid (E300) and salt derivatives (E301-303) are commonly used as food additives, with the additional advantage of their antioxidant and flavor enhancing properties. In wine industry, L-ascorbic acid can be added to prevent oxidation of wine. In beer production, L-ascorbic acid and its salts are efficient anti-oxidants and have a positive influence on aroma, taste and stability. In the meat industry, L-ascorbic acid is used as a reddening agent and as an anti-oxidant. L-ascorbic acid is also used in the pharmaceutical industry and as additive in animal feed. If L-ascorbic acid is added in food production, the content must be controlled.
Citric acid is a very important metabolite in animals, plants and micro-organisms. It is the best known acid of the fruit acids and is contained in a high amount e. g. in citrus fruits (approx. 10 g citric acid/l orange juice). Citric acid is manufactured biotechnologically on a large scale as an additive for food (E330). Citric acid is used as preservative (acidifier) for food or cosmetics, as a chelating agent for metals (e.g. for iron in wine), as an emulsifier (e. g. in the production of processed cheese), and as flavoring agent in the production of soft drinks and candies. It is also a component of many pharmaceuticals and washing powders.
Formic acid occurs as a metabolite in many biochemical reactions, but its concentration is invariably very low. It is the oxidation product of methanol and formaldehyde. As formic acid in low concentrations has both a bactericidal and fungicidal effect, it can be used as a food preservative. Molds tend to produce formic acid as a metabolite; hence, determination of formic acid can give an indication as to the properties, e. g. the degree of decomposition, of samples. Formic acid belongs to the dangerous substances in the production of yeast. Formic acid is a side product in acetic acid fermentation in vinegar production, and is also a part of the “volatile acids” in wine.
Gluconic acid occurs naturally in fruit, honey and wine, but also in meat or dairy products. As a food additive (E574), it is an acidity regulator. It is also used in cleaning products where it dissolves mineral deposits. This acid imparts a sour but refreshing taste. For the wine industry, Botrytis infected grapes exhibit increased levels of this acid that can reach 1-2 g/L. D-Glucono-δ-lactone is found in association with D-gluconic acid, e.g. in wine, and is also widely used in the food industry (yogurt, cottage cheese, bread, confectionery, meat and sausages) in order to control the acidity.
L-glutamic acid (L-glutamate), one of the 20 common amino acids, occurs naturally in foods such as cheese, milk, meat, fish, corn, tomatoes, mushrooms, soybean and sugar beet. The knowledge of the L-glutamate content is of interest e. g. in the ripening of plants, in liver used for the production of human food, and in the control of ripening of cheese. L-glutamic acid is one of the major flavor enhancing components of food (Monosodium glutamate; MSG). Glutamate is an allergen; excessive use of MSG as a food additive (E621) may result in the symptoms of “Chinese Restaurant Syndrome”.
An increase in the content of D-3-hydroxybutyric acid in eggs can be detected 6 days after chickens have been fertilized. This increase continues even after the embryo has died. D-3-Hydroxybutyric acid is thus a typical indicator for fertilized and incubated eggs up to 10 mg/kg (legal limit), and can reach as much as 800 mg/kg in badly spoiled eggs. In egg and bakery products, the level of hydroxybutyric acid is an indicator on how fresh the eggs were when they were used.
D-Isocitric acid is part of the citric acid cycle, and hence occurs in all animals and plants. The contents are usually very low. The determination of D-isocitric acid has become of importance in the analysis of fruit juices (especially of orange juice) for the detection of illegal additives (e. g. citric acid). Not only the minimum content of D-isocitric acid in juices is of interest, but also the ratio citric acid to D-isocitric acid: A ratio which is too high indicates the addition of citric acid. In authentic orange juice, for example, the ratio of citric acid to D-isocitric acid is usually less than 130 and a higher value may be indicative of fruit juice adulteration. The content of D-isocitric acid in products containing orange juice may also be used to calculate its content, e. g. in soft drinks that declare on the bottle label “contains orange juice”.
D-Lactate is very often measured together with L-lactate, and almost never alone. D-Lactic acid is formed only by some micro-organisms, e.g. from Lactobacillus lactis, Lb. bulgaricus and Leuconostoc cremoris. D-Lactic acid is not formed or only in traces by “higher organisms”, e.g. by animals. Therefore the presence of D-lactate may serve as an indicator for microbial contamination or spoilage, presumed that fermentation techniques have not been used in the production of the foodstuff.
D-lactate is very often measured together with L-lactate, and almost never alone. D-Lactic acid is formed by some micro-organisms only, e. g. from Lactobacillus lactis, Lb. bulgaricus and Leuconostoc cremoris. D-lactic acid is not formed or only in traces by “higher organisms”, e. g. by animals. Therefore the presence of D-lactate may serve as an indicator for microbial contamination or spoilage, presumed that fermentation techniques have not been used in the production of the foodstuff, for example for the production of sour milk products.
L-Lactic acid is found in many foods and beverages. Produced naturally by lactic acid bacteria, L-lactic acid is found in many fermented milk products such as yoghurt, and also in pickled vegetables, cured meats and fish. It is commonly supplemented into foods and beverages (E270) as a non-volatile acidulant. In the wine industry, the course of malolactic fermentation is monitored by following the falling level of L-malic acid, and the increasing level of L-lactic acid. The content of L-lactate in beer indicates the presence of Lactobacilli in production. The content of L-lactate in liquid whole egg or in egg powder gives good information about the hygienic situation of the products. Similarly, the quality of milk and fruit and vegetables can be established by measurement of L-lactic acid content. L-Lactate in milk powder indicates the use of neutralized sour milk for the production of milk powder. L-Lactate in wine is also formed during the “second fermentation” (malo-lactic fermentation).
D-malic acid practically does not occur in nature; it is a metabolite produced only by some micro-organisms. Analysis of freshly pressed fruit juices shows the presence of D-malic acid at the detection limit. Hence, the legal limits for fruit and fruit juices are recommended as being 10 mg/l. D-malic acid is a component of the (racemic) D-/L-malic acid when prepared chemically. As natural products are practically free from D-malic acid, the detection of D-malic acid indicates that D-/L-malic acid has been added e. g. to wine or to fruit juice, which may be allowed or forbidden depending on the country.
L-malic acid is one of the most important fruit acids, and the quantitative determination of L-malic acid is especially important in the manufacture of wine, beer, bread, fruit and vegetable products, as well as in cosmetics and pharmaceuticals. Microbial decomposition of L-malic acid leads to the formation of L-lactic acid, which can be a desirable reaction in the production of wine (malo-lactic fermentation, biological de-acidification), or an undesirable reaction in the case of beer (second fermentation). L-malic acid also finds many applications as a food preservative (E296) because it is a stronger acid than citric acid and as flavor enhancing compound.
Oxalic acid may occur in high concentrations in plant leaves. Oxalate in food influences the level of oxalate in urine, and under pathological situations its presence can lead to the formation of calcium oxalate stones in the ureter and kidneys. Furthermore, oxalate in food interferes with resorption and hence availability of calcium, and is thus of considerable nutritional importance.
Spinach, rhubarb, peanuts, chocolates, parsley and tea contain high levels of oxalate. Foodstuffs containing high amounts of oxalate should not be included in the diet of growing children (spinach!), pregnant women, persons with calcium deficiency and patients who have passed a calcium oxalate stone.
Oxalic acid in beer, especially calcium oxalate, is responsible for the gushing effect, for turbidities and precipitates.
As a metabolite of the citric acid cycle, succinic acid occurs widely in animals, plants and micro-organisms. Succinic acid is a specific indicator of microbial decomposition in eggs and egg products (> 5 mg/kg). Succinic acid concentration is also monitored in the manufacture of numerous foodstuffs and beverages, including wine, soy sauce, soy bean flour, fruit juice and dairy products (e.g. cheese), and is also used as a flavoring agent. The ripening process of apples can be followed by monitoring the falling levels of succinic acid. Succinic acid also finds use in many non-food applications (dyes, drugs, perfumes, lacquers, coolants).
Tartaric acid is one of the main acids in grapes and therefore an important parameter in wine production.
Sugar determination
Enzymatic tests can quickly and precisely determine the content of various sugars such as glucose, fructose, lactose, sucrose, and maltose in products ranging from beverages and dairy products to dietary foods and confectionery. Accurate sugar determination is vital for product quality and compliance with nutritional standards. Click on one of the following sugars to learn more about its relevance in food production.
β-Glucan is a carbohydrate with high molecular weight consisting of linear and branched glucose polymers. At high concentrations, it forms so-called “gels” or “films“ that can block the filters used in breweries. ß-Glucan is formed during the production of malt (from barley), but should not exceed a maximum level before the malt is sold to the breweries. Therefore, it is the second most important parameter tested in the malt industry, with specific methods that are registered only for barley and malt. ß-Glucan is also present in other plants like fungi and grapes, but with a molecular structure that is specific for each plant so the tests developed for barley and malt cannot be used.
Galactose is a monosaccharide sugar, one molecule is linked with one glucose molecule and forms a lactose molecule. Therefore, galactose is mainly found in milk and milk products as lactose disaccharide. However, the monosaccharide can also be present in free form, not in the milk itself but in milk products like cheese. D-Galactose can also be found in fruit and vegetables and in polymeric form in legumes as stachyose and raffinose. In lactose-free dairy products, galactose and glucose are formed in their free form through the enzymatic cleavage of lactose. As a component of breast milk, it is an important source of energy for infants. The galactose is absorbed in the small intestine and either broken down in the liver by the enzyme galactosidase, or incorporated into glycogen by uridine diphosphate glucose.
- Enzytec™ Liquid D-Galactose (Art. No. E8120)
- RIDA®CUBE D-Galactose (Art. No. RCS4120)
- Enzytec™ Multi-sugar standard low & high (Art. No. E8440 / E8445)
D-glucose occurs widely in animal and plant kingdoms. It is an essential component of carbohydrate metabolism and occurs frequently in free form along with D-fructose and sucrose. However, the more important forms are those of di-, tri-, oligo- and polysaccharides (lactose, maltose, sucrose; Raffinose, dextrins and starch, cellulose). In food, it is present in significant quantities in honey, wine, beer and fruit juices, and in a range of solid foodstuffs such as bread and pastries, chocolate and candies. The determination of glucose is particularly important in dietary products.
D-glucose and D-fructose are found in most plant products. In food, they are present in significant quantities in honey, wine and beer, and a range of solid foodstuffs such as bread and pastries, chocolate and candies. Fructose, or fruit sugar, is a monosaccharide found in many plants, where it is often bonded to glucose to form the disaccharide sucrose. In the beer and wine industry, the sum of D-glucose and D-fructose is a key parameter, as it represents the amount of sugar that is available to the yeast for conversion into ethanol. In honey, the ratio between both sugars allows detecting an un-allowed addition of sugar like glucose syrup. D-glucose and D-fructose are measured in many food samples and can be measured either independently (like for honey) or simultaneously (like for wine).
Lactose is a disaccharide sugar composed of galactose and glucose. Lactose is an important carbohydrate component of mammalian milk; it is therefore of nutritional importance. The lactose concentration of milk from healthy cows is approx. 4.6 to 5 g /100 g. Lactase (ß-galactosidase), an enzyme found in gastric juice, splits down lactose into D-galactose and D-glucose. People who lack this enzyme after childhood cannot digest milk and are said to be lactose intolerant. Determination of lactose concentrations is particularly important for food products labelled as lactose-free. Moreover, lactose can be used as a sweetening agent in the food industry and should therefore be controlled. D-galactose occurs very seldom in free form; therefore the measurement over the galactose moiety (lactose/galactose test) is more reliable than over the glucose moiety (lactose/glucose test).
Lactose is a disaccharide composed of D-galactose and D-glucose and present in cow milk at a concentration of approx. 50 g/L. D-Galactose occurs very seldom in the free form and is not present in milk. Therefore, the measurement over the galactose moiety (lactose/galactose test) is more reliable than over the glucose moiety (lactose/glucose test) when testing food products manufactured with normal milk or without any milk.
When producing lactose-free milk, the lactose disaccharide is hydrolysed by adding the enzyme lactase (ß-galactosidase), resulting into D-galactose and D‑glucose with approx. 25 g/L each. Lactose/galactose testing is impossible because of the large excess of galactose (25 g/L) that cannot be removed. For this reason, it is mandatory to remove the glucose excess (25 g/L) by using glucose oxidase and then to test the sample with the lactose/glucose test. R-Biopharm AG offers a separate test kit for removing the glucose excess (EnzytecTM Glucose Remover, Art. No. E3400).
- Enzytec™ Liquid Lactose / D-Glucose (Art. No. E8130)
- RIDA®CUBE Lactose / D-Glucose (Art. No. RCS4130)
- Enzytec™ Glucose remover (Art. No. E3400)
- Enzytec™ Multi-sugar standard low & high (Art. No. E8440 / E8445)
Malting grains develops the enzymes required for modifying the starch from the grain into various types of sugar, including the monosaccharide glucose, the disaccharide maltose, the trisaccharide maltotriose, and higher sugars called maltodextrins. Glucose syrup (starch sugar) produced from starch by means of amyloglucosidase also contains maltose. Maltose is measured together with sucrose and D-glucose, because the enzyme α-glucosidase not only reacts with maltose but also with sucrose.
Raffinose is a trisaccharide composed of galactose, glucose and fructose. It can be found in beans, cabbage, brussels sprouts, broccoli, asparagus, other vegetables and whole grains. Raffinose can be hydrolyzed to D-galactose and sucrose by the enzyme α-galactosidase (α-GAL), an enzyme not found in the human digestive tract. Humans and other monogastric animals (pigs and poultry) do not possess the α-GAL enzyme, and these oligosaccharides pass undigested through the stomach and upper intestine. In the lower intestine, they are fermented by gas-producing bacteria that do possess the α-GAL enzyme and make carbon dioxide, methane or hydrogen—leading to the flatulence commonly associated with eating beans and other vegetables.
Starch is a polymeric carbohydrate consisting of a large number of glucose units joined by α-1,4- and α-1,6-glycosidic bonds (amylose, amylopectin). This polysaccharide is produced by most green plants as an energy store. It is the most common carbohydrate in human diets and is contained in large amounts in staple foods such as potatoes, wheat, maize (corn) and rice. Starch is an important ingredient of food and animal feed. It serves as binder e. g. in sauces, filler, thickener and texturizer in the production of various foodstuffs. Partially hydrolyzed starches are also used in soft drinks, and illegally added to wine to increase the dry extract (and thereby pretend a better wine quality).
Sucrose and glucose have a central position in plant metabolism. In food, they are present in significant quantities in honey, wine, beer and fruit juices, and in a range of solid foodstuffs such as bread and pastries, chocolate and candies. Sucrose is also known as table sugar, and its isolation from cane and beet is of great economic interest. When sucrose is hydrolyzed (inverted), D-glucose and D-fructose are formed. Sucrose is an important ingredient of foodstuffs, not only as sweetener but also giving monetary value. Specific sugar ratios exist for many fruits and for honey; this can be used as an indication of added sugar.
Sucrose, D-glucose and D-fructose are found in most plant and food products. In plant materials, D-glucose and D-fructose occur as free sugars and in a range of oligo- and polysaccharides such as fructans (inulins), starch, and cellulose. In food, they are present in significant quantities in honey, wine, beer and fruit juices, and in a range of solid foodstuffs such as bread and pastries, chocolate and candies.
Other parameters
Beyond acids and sugars, enzymatic testing can determine a variety of other parameters, including alcohols, salts, sulfite, cholesterol, ammonia, and metals such as copper and iron. These tests are crucial for ensuring comprehensive quality control and safety in food production.
Acetaldehyde occurs naturally in all organisms, even if in very small quantities, because it is a product of many metabolic processes. Acetaldehyde occurs naturally in coffee, bread and ripe fruit, and is produced by plants. In food production, the concentration of acetaldehyde rises considerably when ethanol fermentation takes place, with as much as 100 mg/l in wine and 20 mg/l in beer. On the other hand, acetaldehyde is an important flavor compound in wine and beer. In dairy products like yogurt and cheese, it is responsible for desirable flavors but also for flavor defects.
During fermentation of grape juice, yeasts require nitrogen as a nutrient, with a portion provided by primary amino acids. The corresponding test can be called Primary Amino Acid Nitrogen (PAAN) or α-Amino Nitrogen. The Yeast Available Nitrogen (YAN) corresponds to the total nitrogen available for the yeast growth, and must be determined by adding the Amonia Nitrogen content (AN) to the PAAN.
Ammonia is present in the environment as a result of natural processes from the nitrogen cycle, but also from industrial activity including intensive farming. High concentrations of ammonia can indicate the (microbial) decomposition of substances like milk, meat and seafood, where it is a major component of the off-flavors and odors associated with spoilage. Ammonia also indicates the presence of feces, urine and microorganisms in water. On the positive side, ammonia is an important source of nitrogen for many microorganisms in fermentation processes like winemaking. Ammonium salts produced on a large scale are used for the production of fertilizers, animal feed, paper, and in the case of food as leavening agents, stabilizers, and for flavoring purposes.
Cholesterol is the most important of the animal sterols. It is an important component of the cell membranes of the higher species, and a precursor to a whole series of steroid hormones. Cholesterol is found in all animal fats and is an important component of egg yolk. It is found in foods such as meat, poultry, seafood and dairy products. Due to the relative constancy of its content, it is frequently used in the determination of the egg content of foodstuffs like bakery goods, noodles and liqueur with egg yolks. A high cholesterol diet can cause heart, liver and kidney diseases. The daily intake should not exceed 300 mg.
Determination of copper content is important in wine production. Excess copper ions, e.g. from fining, can cause formation of an unsightly haze in finished wine.
Ethanol occurs in nature in practically all organisms, even if in very small quantities. It is the end product of alcoholic fermentation and a desired component of alcoholic beverages, but also the undesirable component in non-alcoholic and low-alcoholic beverages, or in other foodstuffs including chocolates, sweets, jam, honey, vinegar and dairy products. The presence of ethanol in fruit products like fruit juices indicates that the components used for production may have decomposed. The presence of ethanol is also an indirect indicator for the presence of yeasts. In meat products, ethanol indicates spoilage. In non-food industry, ethanol is a solvent e. g. for essential oils and pharmaceutical substances.
Glycerol, a by-product of alcoholic fermentation, and its fatty acid esters (glycerides) occur frequently in nature. In the food industry, glycerol is an important moistening agent for baked goods. It is also added to candies and icings to prevent crystallization, as a solvent for food colors and a carrier for extracts and flavoring agents. As a product of fermentation, glycerol is monitored in the beer and wine industries, where it occurs at concentrations of approx. 1 % (v/v). Glycerol contributes to the aroma; its sweet taste brings the wine “body”. In the pharmaceutical industry, the smoothness of lotions, creams and toothpaste is due to the incorporation of glycerol. The glycerol backbone is found in all lipids known as triglycerides.
Implicated in haze formation (along with copper), iron ions can contaminate wine through contact with exposed iron surfaces (non-stainless steel). The maximum value is around 8 mg/l; above this limit the danger of iron haze is high.
The nitrogen required by plants for growing (formation of proteins) is taken up by plants almost entirely in the form of nitrate (fertilizers). Nitrate in foodstuffs is of nutritional importance due to its reduction to nitrite and the formation of compounds that attach to hemoglobin. It also forms nitrosamines that are known to be carcinogenic. Some plants, e. g. cabbage, red beets, radish, spinach and salad have the ability to store nitrate in their tissue. The content of nitrate in potatoes is relatively low. When cooking these vegetables, a lot of nitrate will be eliminated in the boiling water. In meat production, potassium nitrate is used as a salting and reddening agent. However, the nitrate/nitrite concentration in meat products must not exceed 100 mg/kg. In fruit juices, high nitrate concentrations indicate that tap water has been added. There are also limits for the concentration of nitrate in drinking / tap water (e.g. 50 mg/l in Europe). “Natural” water contains approx. 1 mg nitrate/l.
D-sorbitol is a sugar alcohol that occurs extensively in fruits, e. g. in apples, cherries, pears, plums, but it is not or only in traces contained in grapes, grape juice and wine. It is a parameter to determine the authenticity of red colored juices and to check the fruit content. For example, the concentration of D-sorbitol may be used for the calculation of the apple juice content of beverages declared as “contains apple juice”. D-sorbitol has an anti-cariogenic influence, but may have a laxative effect if large quantities are consumed (10 – 50 g per day). It is used in the food technology industry as a moisturizing agent and as a sugar substitute (E420) for diabetic products because insulin is not necessary for metabolism. D-sorbitol is stable to acids, improves the texture of food and is a non-browning component. Xylitol is a sugar alcohol that occurs frequently in fruits, vegetables and mushrooms. Xylitol is produced on an industrial scale. Xylitol is not fermented by cariogenic bacteria (e. g. of Streptococcus mutans) in the mouth.
Sulfur dioxide in a wine barrel is in equilibrium between different forms ranging from SO2 (which is a gas present in traces), to free SO2 (5 – 50 mg/L) and bound SO2 (100 – 400 mg/L). The bound SO2 is not active against wine spoilage and oxidation, so it is mandatory to monitor the level of free SO2 with a specific test. The total SO2 is the sum of free and bound SO2.
Sulfur dioxide (SO2), sulfurous acid (H2SO3) and sulfites (SO3 salts) occur in very low concentrations in nature. However, sulfites are widely used as preservatives (E220 to E228) in the food industry, in order to prevent microbial spoilage and oxidation. The usage of sulfite (SO3) and sulfur dioxide (SO2) belongs to the most important techniques in order to improve stability and taste of wine.
Sulfite is regarded as being poisonous for cells. Its content is legally limited for specific foodstuffs in a number of countries, and the content has often to be declared on the label because of sulfite intolerance in some individuals. As example, a limit value of 10 mg/L exists for juices.
Urea is the most important decomposition product of protein metabolism. Measurement of urea in body fluids gives an indication of the state of protein balance in muscle cells and of the protein supply e. g. of cows. Urea is sometimes added (illegally) to meat products in order to indicate a higher content of muscle protein than is actually present (the addition of 1 % urea pretends the addition of approx. 3 % of protein). Moreover, urea is an indicator for the presence of urine in swimming pools. It is also used as a component in the manufacture of cosmetics, pharmaceuticals and paper.
Product overview
You can find a comprehensive overview of our test kits for enzymatic analysis in our free brochure.
Key benefits of monitoring food constituents
- Accurate ingredient analysis: Ensures precise measurement of sugars, acids, alcohols, and other food components, maintaining product quality and consistency.
- Compliance with standards: Helps manufacturers meet international standards and regulations, such as those set by AOAC, CEN, IDF, IFU, ISO, and OIV.
- Quality control: Enhances the quality control process, ensuring that products meet the desired specifications and consumer expectations.
- Versatility: Suitable for a wide range of food products, including fruit juices, wine, beer, dairy products, eggs, and meat, making it a valuable tool for various segments of the food industry.
- Ease of automation: Facilitates automation in laboratories, improving efficiency and accuracy in routine analysis.
Conclusion
Monitoring food constituents is essential for maintaining the quality, safety, and compliance of food products. Using advanced enzymatic assays and technologies such as the SureFood® and Enzytec™ product lines, manufacturers can achieve precise and reliable measurement of key food components. This not only ensures product consistency and consumer satisfaction but also helps meet stringent international standards and regulations. Implementing comprehensive constituent testing supports robust quality control and enhances overall production efficiency.
Product portfolio
| Product | Description | No. of tests/amount | Art. No. |
|---|---|---|---|
| Enzytec™ Liquid D-3-Hydroxybutyric acid |
Enzymatic assay for D-3-hydroxybutyric acid/hydroxybutyrate in foodstuff and other sample materials. Read more |
Test-kit with 50 determinations for manual use, 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8540 |
| Enzytec™ Liquid Combi Maltose/Sucrose/D-Glucose |
UV assay for the determination of maltose, sucrose and D-glucose in foodstuffs and other sample materials. Read more |
25 determinations each 1 x 50 mL R1; 1 x 50 mL R2; 1 x 50 mL R3; 1 x 37.5 mL R4 |
E8175 |
| Enzytec™ Liquid Succinic acid |
Enzymatic UV assay for the determination of succinic acid in foodstuffs and other sample materials. Read more |
Test-kit with 25 determinations for manual use 1 x 50 ml R1 and 1 x 12.5 ml R2 |
E8580 |
| Enzytec™ Sodium |
Enzytec™ Sodium is an enzymatic assay for the determination of sodium in foodstuff and other sample materials. Read more |
Test kit for 400 tests on automatic analyzer Reagents: # 1: Buffer (> 0.050 mol/L): 4 bottles of approx. 20 mL # 2: R2 – GAL (GAL < 50 KU/L): 4 bottles of approx. 20 mL # 3: R3 – DIL (Diluent < 0.2 mol/L): 4 bottles of approx. 8.5 mL # 4: R4 – ONPG (ONPG < 50 mmol/L): 2 bottles of approx. 8.5 mL # 5: Liquid Standard 1: 1 vial of approx. 5 mL (Na+ = 2.759 g/L) # 6: Liquid Standard 2: 1 vial of approx. 5 mL (Na+ = 4.138 mg/L) |
E2590 |
| Enzytec™ Alpha-amino Nitrogen |
Enzytec™ Alpha-amino Nitrogen is a colorimetric assay for the determination of alpha-amino nitrogen in foodstuff and other sample materials. Read more |
Test kit for 75 determinations # 1: R1 – Buffer pH > 7.00: 3 bottles with approx. 50 mL # 2: R2 – OPA > 5 mM: 1 bottle with approx. 16 mL # 3: R3 – CAL: 1 bottle with approx. 5 mL (solution of α-amino nitrogen = 150 mg/L, NaN3 < 0.1%) |
E2500 |
| Enzytec™ Total Protein |
Enzytec™ Total Protein is an colorimetric assay for the determination of Total Protein in foodstuff and other sample materials. Read more |
Test kit for 105 determinations #1: R1 – Chromogen (Pyrogallol Red), < 2 μmol/L: 3 bottles approx. 70 mL #2: R2 – CAL (Protein solution = Concentration value shown on the label, NaN3 < 0.1%): 1 bottle approx. 5 mL |
E2620 |
| Enzytec™ Polyphenols |
Enzytec™ Polyphenols is a colorimetric assay for the determination of Total Polyphenols in foodstuff and other sample materials. Read more |
Test kit for 80 determinations # 1: Folin reagent modified: 2 bottles of 80 mL # 2: Sodium carbonate >10 g/L: 1 bottle of 80 mL # 3: Sodium hydroxide >1 g/L: 2 bottles of 40 mL # 4: Standard liquid: Solution of gallic acid 3 g/L, NaN3 < 0.1%, 1 bottle of 5 mL |
E2530 |
| Enzytec™ Anthocyanins |
Enzytec™ Anthocyanins is a colorimetric assay for the determination of Anthocyanins in foodstuffs and other sample materials. Read more |
Test kit for 100 determinations # R1: Buffer (5 x 40 mL); Modified halogenuric buffer, 100 mM, pH < 2; Stabilizers; NaN3 < 0.1% |
E2510 |
| Enzytec™ Chloride |
Enzytec™ Chloride is a colorimetric assay for the determination of Chloride in foodstuffs and other sample materials. Read more |
Test kit for 200 determinations Reagents: # 1: Mercury thiocyanate 2.2 mmol/L, Ferric nitrate 20 mmol/L; 5 bottles of approx. 80 mL # 2: Reagent Blank: approx. 20 mL # 3: Standard Chloride, 4.0 g/L (113 mmol/L), NaN3 < 0.1%, approx. 5 mL |
E2520 |
| Enzytec™ Pyruvate |
Enzytec™ Pyruvate is a UV assay for the determination of Pyruvate in foodstuff and other sample materials. Read more |
Test kit for 50 determinations #1: R1 – Buffer (> 400 mmol/L): 2 vials of approx. 60 mL #2: R2 – NADH (LDH > 250 KU/L): 5 vials of approx. 20 mL #3: R3 – LDH Ready to use (NADH > 0.1 mmol/L): 1 vial of approx. 2.5 mL #4: R4 – Liquid Standard: 1 vial of approx. 10 mL (pyruvate solution = 400 mg/L) |
E2550 |
| Enzytec™ Potassium |
Enzytec™ Potassium is an enzymatic assay for the determination of Potassium in foodstuff and other sample materials. Read more |
Test kit for 400 determinations on automatic analyzer # 1: Buffer (> 0.050 mol/L): 4 x approx. 20 mL # 2: R2 – PK (PK > 50 KU/L): 4 vials give approx. 20 mL each # 3: R3 – LDH ready to use (< 300 KU/L): 2 x approx. 8.5 mL # 4: Liquid Standard 1: approx. 5 mL (K+ = 97.75 mg/L) # 5: Liquid Standard 2: approx. 5 mL (K+ = 293.25 mg/L) |
E2540 |
| RIDA®CUBE Citric acid |
UV-method for the determination of Citric acid in food products. The enzymatic test kit is designed for using only with the RIDA®CUBE SCAN instrument (340 nm). Read more |
Test-kit for 32 determinations (single-test cartridges) | RCS4230 |
| Enzytec™ Liquid Acetaldehyde |
Enzymatic assay for Acetaldehyde in foodstuff and other sample materials. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8300 |
| Enzytec™ Liquid Multi-acid standard 2 low & high |
The Enzytec™ Liquid Multi-acid standard 2 low (E8470) is intended to be used as a quality-control sample for determinations with the Enzytec™ Liquid enzymatic kits. It is a stable aqueous solution to control precision and correctness during … Read more |
E8470 = 3 x 3.5 mL E8475 = 3 x 3.5 mL |
E8470 / E8475 |
| Enzytec™ Liquid L-Glutamic acid |
Enzymatic UV test for the determination of L-glutamic acid in foodstuffs and other sample materials. Read more |
Test-kit with 2 x 25 determinations for manual use, 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8530 |
| Enzytec™ Liquid Cholesterol |
Enzymatic assay for Cholesterol in foodstuff and other sample materials. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8320 |
| Enzytec™ Liquid Nitrate |
Enzymatic assay for Nitrate in foodstuff and other sample materials. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8370 |
| Enzytec™ Liquid D-Malic acid |
Enzymatic assay for D-Malic acid in foodstuff and other sample materials. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8270 |
| Enzytec™ Liquid Formic acid |
Enzymatic assay for Formic acid in foodstuff and other sample materials. Read more |
Test-kit with 25 determinations for manual use, (500 tests on automated systems), 1 x 50 ml R1 and 1 x 12.5 ml R2 |
E8510 |
| Enzytec™ Liquid Starch |
Enzymatic assay for the determination of native starch in foodstuff and other sample materials. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8100 |
| Enzytec™ Liquid Maltose/Sucrose/D-Glucose |
Enzymatic assay for maltose, sucrose and D-glucose in foodstuff and other sample materials. Read more |
Test-kit with 50 determinations for manual use, 2 x 50 mL R1 and 2 x 12.5 mL R2 |
E8170 |
| Enzytec™ Liquid D-Lactic acid |
Enzymatic assay for D-Lactic acid in foodstuff and other sample materials. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8245 |
| Enzytec™ Liquid D-Gluconic acid |
Enzymatic assay for the determination of D-Gluconic acid in foodstuff and other sample materials. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8520 |
| Enzytec™ Liquid Urea/Ammonia |
Enzymatic assay for Urea and Ammonia in foodstuff and other sample materials. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8395 |
| Enzytec™ Generic D-Isocitric acid |
UV-method for the determination of D-Isocitric acid in foodstuff and other materials. Read more |
Test combination for approx. 32 determinations | E1222 |
| Enzytec™ Generic Sulfite (SO2-Total) |
UV-method for the determination of Sulfite (“total SO2“) in foodstuff and other materials. Read more |
Test combination for approx. 30 determinations | E6275 |
| Enzytec™ Generic D-3-Hydroxybutyric acid |
UV-method for the determination of D-3-Hydroybutyric acid in foodstuff and other materials. Read more |
Test combination for approx. 33 determinations | E2610 |
| Enzytec™ Liquid Multi-acid standard low & high |
The Enzytec™ Liquid Multi-acid standard low (E8460) is intended to be used as a quality-control sample for determinations with the Enzytec™ Liquid enzymatic kits. It is a stable aqueous solution to control precision and correctness during manual … Read more |
E8460 = 3 x 3.5 mL E8465 = 3 x 3.5 mL |
E8460 / E8465 |
| Enzytec™ Multi-sugar standard low & high |
The Enzytec™ Multi-sugar standard low (E8440) is intended to be used as a quality-control sample for determinations with the Enzytec™ enzymatic kits. It is a stable aqueous solution to control precision and correctness during manual assays. This … Read more |
E8440 = 3 x 3.5 mL E8445 = 3 x 3.5 mL |
E8440 / E8445 |
| Enzytec™ Liquid Glycerol |
Enzymatic assay for Glycerol in foodstuff and other sample materials. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8360 |
| RIDA®CUBE SO2-Free |
UV-method for the determination of free sulfite in wine, must and other food products. The enzymatic test kit is designed for using only with the RIDA®CUBE SCAN instrument (340 nm). Read more |
Test-kit for 32 determinations (single-test cartridges) | RCS4610 |
| Enzytec™ Liquid Ammonia |
Enzymatic assay for Ammonia in foodstuff and other sample materials. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8390 |
| RIDA®CUBE Ammonia |
UV-method for the determination of Ammonia (NH4) in food products and other sample materials. The enzymatic test kit is designed for using only with the RIDA®CUBE SCAN instrument (340 nm). Read more |
Test-kit for 32 determinations (single-test cartridges) | RCS4390 |
| RIDA®CUBE Lactose / D-Glucose |
UV-method for the determination of Lactose / D-Glucose (without differentiation) in food products and other sample materials. The enzymatic test kit is designed for using only with the RIDA®CUBE SCAN instrument (340 nm). Read more |
Test-kit for 32 determinations (single-test cartridges) | RCS4130 |
| Enzytec™ Liquid Acetic acid |
Enzymatic assay for the determination of Acetic acid in foodstuff and other sample materials. AOAC® Official Method℠ 2024.01 for wines, juices, sauces/remoulades, kombucha, beer, sausages/meat, vinegar, and microbiological culture media. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8226 |
| RIDA®CUBE D-Galactose |
Enzymatic method for the determination of D-Galactose in food products and other sample materials (UV-method). This test-kit is designed for using only with the RIDA®CUBE SCAN instrument (340 nm). Read more |
Test-kit for 32 determinations (single-test cartridges) | RCS4120 |
| RIDA®CUBE Lactose / D-Galactose |
Enzymatic method for the determination of Lactose / D-Galactose (without differentiation) in food products and other sample materials (UV-method). This test-kit is designed for using only with the RIDA®CUBE SCAN instrument (340 nm). Read more |
Test-kit for 32 determinations (single-test cartridges) | RCS4110 |
| Enzytec™ Liquid D-Galactose |
Enzymatic assay for D-Galactose in foodstuff and other sample materials. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8120 |
| Enzytec™ Liquid Lactose / D-Galactose |
Enzymatic assay for Lactose / D-Galactose in foodstuff and other sample materials (without differentiation). Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8110 |
| RIDA®CUBE Ethanol |
UV-method for the determination of Ethanol in food products. The enzymatic test kit is designed for using only with the RIDA®CUBE SCAN instrument (340 nm). Read more |
Test-kit for 32 determinations (single-test cartridges) | RCS4340 |
| Enzytec™ Liquid L-Malic acid |
Enzymatic assay for L-Malic acid in foodstuff and other sample materials. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8280 |
| Enzytec™ Liquid L-Lactic acid |
Enzymatic assay for L-Lactic acid in foodstuff and other sample materials. AOAC® Official Method℠ 2024.07 for wine, milk and fermented milk products, fermented vegetable products, fruit and vegetable juices, beer, eggs, and egg powder. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8260 |
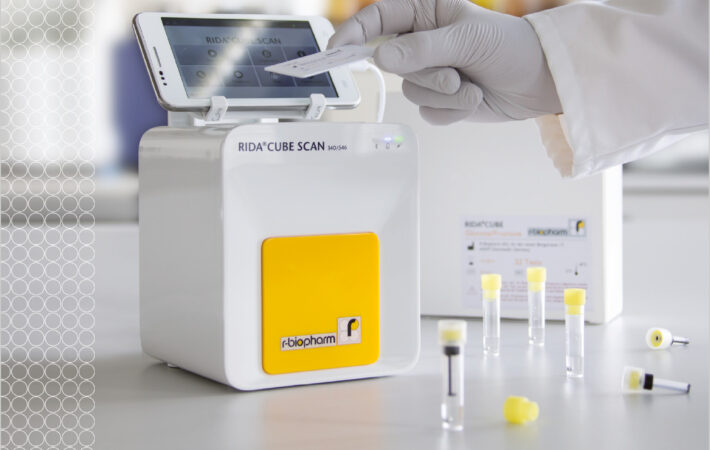
| RIDA®CUBE SCAN |
The RIDA®CUBE SCAN is a photometric system that allows biochemistry testing, covering all enzymatic and colorimetric assays for the detection of organic acids (e.g. lactic acid), sugars (e.g. glucose) or other food components (e.g. sulfite). The … Read more |
Weight: 2.4 kg Dimensions: 16 x 13 x 14.5 cm Android based app Bluetooth and USB connection Data transfer to host printer |
ZRCS0546 |
| RIDA®CUBE SO2-Total |
UV-method for the determination of SO2-Total (free and bound sulfite) in wine, must and other food products. The enzymatic test kit is designed for using only with the RIDA®CUBE SCAN instrument (340 nm). Read more |
Test-kit for 32 determinations (single-test cartridges) | RCS4600 |
| Enzytec™ Liquid Ethanol |
Enzymatic assay for Ethanol in foodstuff and other sample materials. AOAC® Official Method℠ 2017.07 for kombucha, juices and alcohol-free beer. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8340 |
| RIDA®CUBE D-/L-Lactic acid |
UV-method for the determination of D-Lactic acid and L-Lactic acid (without differentiation) in foodproducts. The enzymatic test kit is designed for using only with the RIDA®CUBE SCAN instrument (340 nm). Read more |
Test-kit for 32 determinations (single-test cartridges) | RCS4240 |
| RIDA®CUBE D-Glucose |
UV-method for the determination of D-Glucose in food products. The enzymatic test kit is designed for using only with the RIDA®CUBE SCAN instrument (340 nm). Read more |
Test-kit for 32 determinations (single-test cartridges) | RCS4140 |
| RIDA®CUBE Sucrose / D-Glucose / D-Fructose |
UV-method for the determination of Sucrose / D-Glucose / D-Fructose (without differentiation) in food products. The enzymatic test kit is designed for using only with the RIDA®CUBE SCAN instrument (340 nm). Read more |
Test-kit for 32 determinations (single-test cartridges) | RCS4190 |
| RIDA®CUBE D-Glucose / D-Fructose |
UV-method for the determination of D-Glucose / D-Fructose (without differentiation) in food products. The enzymatic test kit is designed for using only with the RIDA®CUBE SCAN instrument (340 nm). Read more |
Test-kit for 32 determinations (single-test cartridges) | RCS4160 |
| RIDA®CUBE Sucrose / D-Glucose |
UV-method for the determination of Sucrose / D-Glucose (without differentiation) in food products. The enzymatic test kit is designed for using only with the RIDA®CUBE SCAN instrument (340 nm). Read more |
Test-kit for 32 determinations (single-test cartridges) | RCS4180 |
| Enzytec™ Liquid Lactose / D-Glucose |
Enzymatic assay for Lactose / D-Glucose in foodstuff and other sample materials (without differentiation). Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8130 |
| Enzytec™ Liquid D-Glucose / D-Fructose |
Enzymatic assay for D-Glucose / D-Fructose in foodstuff and other sample materials. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 + 2 x 12.5 ml R2 + 2 x 12.5 ml R3 |
E8160 |
| Enzytec™ Liquid D-Glucose |
Enzymatic assay for D-Glucose in foodstuff and other sample materials. AOAC® Official Method℠ 2024.03 fruit- and juices, wine and beer. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8140 |
| Enzytec™ Liquid SO2-Total |
This test is a colorimetric assay used in wine laboratories for the determination of “total sulfite” (SO2 total) concentration in wine, must and other food samples. The assay does not involve any enzyme, the reaction is a chemical / … Read more |
Test-kit with 2 x 50 determinations, 2 x 100 mL R1 + 2 x 25 mL R2 + 3.5 mL calibrator (100 assays) |
E8600 |
| Enzytec™ Liquid Sucrose / D-Glucose |
Enzymatic assay for Sucrose / D-Glucose in foodstuffs and other sample materials (without differentiation). AOAC® Official Method℠ 2024.05 for wine, beer, juices, chocolate, ice cream, sweetened condensed milk, jam and molasses. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8180 |
| Enzytec™ Liquid Sucrose / D-Glucose / D-Fructose |
Enzymatic assay for Sucrose / D-Glucose / D-Fructose (total sugars) in foodstuffs and other sample materials. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8190 |
| Enzytec™ Liquid SO2-Free |
This test is a colorimetric assay used in wine laboratories for the determination of “free sulfite” (SO2 free) concentration in wine, must and other food samples. The assay does not involve any enzyme, the reaction is a chemical / colorimetric … Read more |
Test-kit with 2 x 50 determinations, 2 x 100 mL R1 + 2 x 25 mL R2 + 3.5 mL calibrator (100 assays) |
E8610 |
| RIDA®CUBE L-Malic acid |
UV-method for the determination of L-Malic acid in food products. The enzymatic test kit is designed for using only with the RIDA®CUBE SCAN instrument (340 nm). Read more |
Test-kit for 32 determinations (single-test cartridges) | RCS4280 |
| RIDA®CUBE Acetic acid |
UV-method for the determination of Acetic acid in food products. The enzymatic test kit is designed for using only with the RIDA®CUBE SCAN instrument (340 nm). Read more |
Test-kit for 32 determinations (single-test cartridges) | RCS4226 |
| RIDA®CUBE L-Lactic acid |
UV-method for the determination of L-Lactic acid in food products. The enzymatic test kit is designed for using only with the RIDA®CUBE SCAN instrument (340 nm). Read more |
Test-kit for 32 determinations (single-test cartridges) | RCS4260 |
| Enzytec™ Liquid D-/L-Lactic acid |
Enzymatic assay for D-Lactic acid and L-Lactic acid in foodstuff and other sample materials (without differentiation). AOAC® Official Method℠ 2024.08 for wine, milk and fermented milk products, fruit and vegetable juices, beer and eggs. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8240 |
| Enzytec™ Generic Oxalic acid |
Colorimetric assay for the determination of Oxalic acid in wine, beer, beverages, honey and other foodstuffs. Read more |
1 x 20 ml for 10 determinations | E2100 |
| Raffinose |
UV-method for the determination of raffinose in foodstuffs and other materials. Read more |
Test combination for 32 determinations | 10428167035 |
| Enzytec™ Generic Starch |
UV-method for the determination of Starch in foodstuffs and other materials. Read more |
Test combination for approx. 32 determinations | E1268 |
| Enzytec™ Generic D-Glucose / D-Fructose / Sucrose |
UV-method for the determination of D-Glucose / D-Fructose / Sucrose in foodstuffs and other materials. Read more |
Test combination for approx. 16 determinations each | E1247 |
| Enzytec™ Generic D-Glucose / Sucrose |
UV-method for the determination of D-Glucose / Sucrose in foodstuffs and other materials. Read more |
Test combination for approx. 16 determinations each | E1246 |
| Enzytec™ Generic D-Glucose / D-Fructose |
UV-method for the determination of D-Glucose / D-Fructose in foodstuffs and other materials. Read more |
Test combination for approx. 32 determinations each | E1245 |
| Enzytec™ Generic Lactose / D-Galactose |
UV-method for the determination of Lactose / D-Galactose in foodstuffs and other materials. Read more |
Test combination for approx. 16 determinations each / approx. 32 determinations of lactose | E1213 |
| Enzytec™ Generic D-Glucose |
UV-method for the determination of D-Glucose in foodstuffs and other materials. Read more |
Test combination for approx. 32 determinations | E1210 |
| Enzytec™ Sample purifier (for Oxalic acid) |
Sample preparation and purification for enzymatic testing. Read more |
For 20 samples | E2250 |
| Enzytec™ Generic L-Ascorbic acid |
UV-method for the determination of L-Ascorbic acid in foodstuffs and other materials. Read more |
Test combination for approx. 24 assays | E1267 |
| Enzytec™ Generic D-/L-Lactic acid |
UV-method for the determination of D-/L-Lactic acid in foodstuffs and other materials. Read more |
Test combination for approx. 32 determinations | E1255 |
| Enzytec™ Generic L-Lactic acid |
UV-method for the determination of L-Lactic acid in foodstuffs and other materials. Read more |
Test combination for approx. 32 determinations | E1254 |
| Enzytec™ Generic Acetic acid |
UV-method for the determination of Acetic acid in foodstuffs and other materials. Read more |
Test combination for 2 x 16 determinations (approx. 32 assays) | E1226 |
| Enzytec™ Generic D-Gluconic acid |
UV-method for the determination of D-Gluconic acid (D-Glucono-δ-lactone) in foodstuffs and other materials. Read more |
Test combination for approx. 32 determinations | E1223 |
| Enzytec™ Generic L-Malic acid |
UV-method for the determination of L-Malic acid in foodstuffs and other materials. Read more |
Test combination for approx. 32 determinations | E1215 |
| Enzytec™ Generic Citric acid |
UV-method for the determination of Citric acid in foodstuffs and other materials. Read more |
Test combination for 3 x 8 determinations (approximately 24 assays) | E1214 |
| Alcohol standard |
This alcohol standard is a stable aqueous solution for assay control and the calibration of automated systems to determine the concentration of ethanol. This product includes 10 reclosable glass vials with 1.5 ml alcohol standard each (300 mg/l). … Read more |
10 x 1.5 ml reclosable glass vials (300 mg/l) | AQ03-015 |
| Enzytec™ Color Tartaric acid |
This test is a colorimetric assay used mainly in wine laboratories for determination of tartaric acid concentration in wine and must. The assay does not involve any enzyme, the reaction is a chemical / colorimetric reaction (therefore it belongs ot … Read more |
2 x 80 ml R1 reagent + 1 x 25 ml R2 reagent (100 assays) | E3100 |
| Enzytec™ Color Copper |
This test is a colorimetric assay used mainly in wine laboratories for determination of the Copper (Cu) concentration in wine. The assay does not involve any enzyme, the reaction is a chemical / colorimetric reaction (therefore it belongs ot the … Read more |
2 x 50 ml (50 assays) | E2400 |
| Enzytec™ Color Iron |
This test is a colorimetric assay used mainly in wine laboratories for determination of the Iron (Fe) concentration in wine and other food samples. The assay does not involve any enzyme, the reaction is a chemical / colorimetric reaction (therefore … Read more |
4 x 100 ml (200 assays) | E2300 |
| Enzytec™ Glucose Remover |
Removal of Glucose excess in samples. Read more |
Reagents for 32 samples | E3400 |
| Enzytec™ Color GlucaTest® |
Quantitative determination of high molecular weight ß-Glucan from barley in malt-mash and wort (EBC 4.16.3, MEBAK 3.1.4.9.2), in combination with sample extraction method EBC 4.5.1. The assay does not involve any enzyme, the reaction is a chemical / … Read more |
• GlucaTest S125 (E3500) = 40 tests (1 x 125 ml solution A + calibrator set), • GlucaTest L500 (E3550) = 160 tests (4 x 125 ml solution A + calibrator set) |
E3500 / E3550 |
| Enzytec™ Liquid D-Isocitric acid |
Enzymatic assay for the determination of D-Isocitric acid in foodstuff and other sample materials. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 1 x 2.0 mL Assay control with D-threo-isocitric acid (0.05 g/L) |
E8550 |
| Enzytec™ Liquid Citric acid |
Enzymatic assay for the determination of Citric acid in foodstuff and other sample materials. Read more |
Test-kit with 2 x 25 determinations for manual use, (500 tests on automated systems), 2 x 50 ml R1 and 2 x 12.5 ml R2 |
E8230 |