Recent news in Food & Feed Analysis
- Home
- /
- Antibiotic residues in honey
Antibiotic residues in honey

September is National Honey Month in the USA. As it marks the end of the honey collection season, it’s a good moment to focus on the natural sweetener – which is not always as natural as one might think. Both the bees and the plants, from which the bees collect their nectar, may have been treated with antibiotics. In this way, antibiotic residues can also be found in honey.
Antibiotic exposure is a major problem in global honey trade. Drug residues are being regularly detected in honey – sometimes in alarming concentrations. But how do drugs get into honey after all? First of all, bee diseases such as foulbrood and varroasis can be treated with antibiotics. In addition, antibiotics can be used to treat plant diseases such as fire blight. By collecting nectar from treated plants, antibiotic residues also get into the honey.
Because of potential health risks, the use of antibiotics in beekeeping is generally banned in the EU. With the exception of streptomycin, no maximum residue limits are defined (zero tolerance). Accordingly, honey of European origin is rarely contaminated with antibiotics. However, the use of antibiotics is legal in some other countries – including South America, where a large part of the global honey supply is produced. Testing for residues is therefore relevant particularly in imported honey.
Honey analysis generally covers the following parameters:
- Authenticity (e.g. foreign enzymes and sugars)
- Quality parameters (e.g. water, sugar and acid content)
- Botanical origin
- Genetically modified organisms
- Microbiology & hygiene (e.g. aerobic count, bacteria, yeasts and molds)
- Pesticides
- Bee repellents
- Antibiotic residues
Relevant parameters in terms of antibiotic residues are streptomycin, chloramphenicol and tetracycline. For all three analytes, R-Biopharm tests are available. The tests for streptomycin and chloramphenicol have recently been revised and now provide a simplified sample preparation and test implementation as well as improved values in sensitivity, recovery and detection limit.
R3104: RIDASCREEN® Streptomycin
- Ready-to-use reagents
- Less pipetting through functionalized microtiter plate
- Shortened incubation time of only 45 minutes
- Detection limit of 2 μg/kg
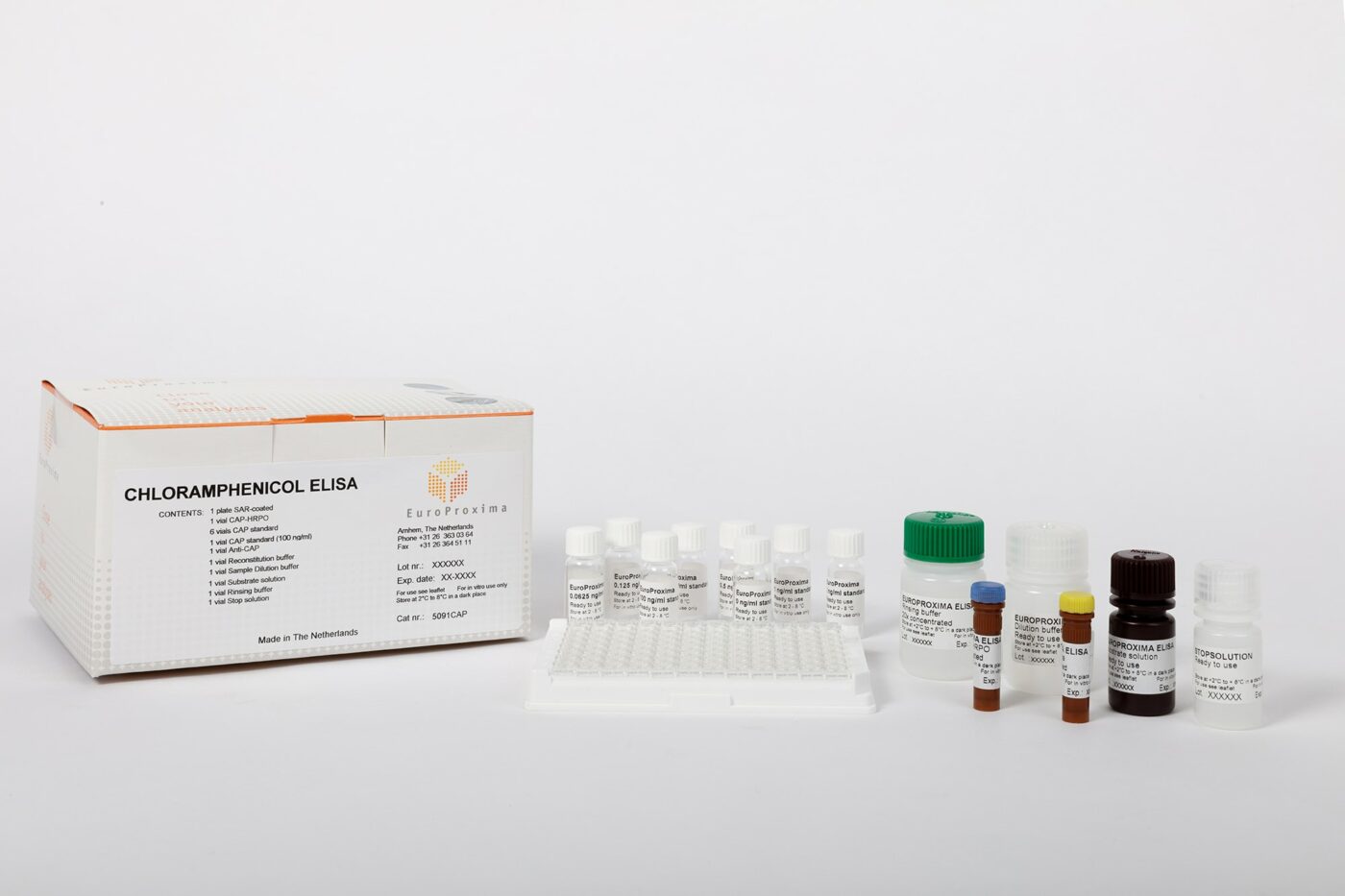
R1511: RIDASCREEN® Chloramphenicol
- Simple sample preparation without IAC or SPE
- Ready-to-use reagents
- Less pipetting through functionalized microtiter plate
- Detection limit below the MRPL
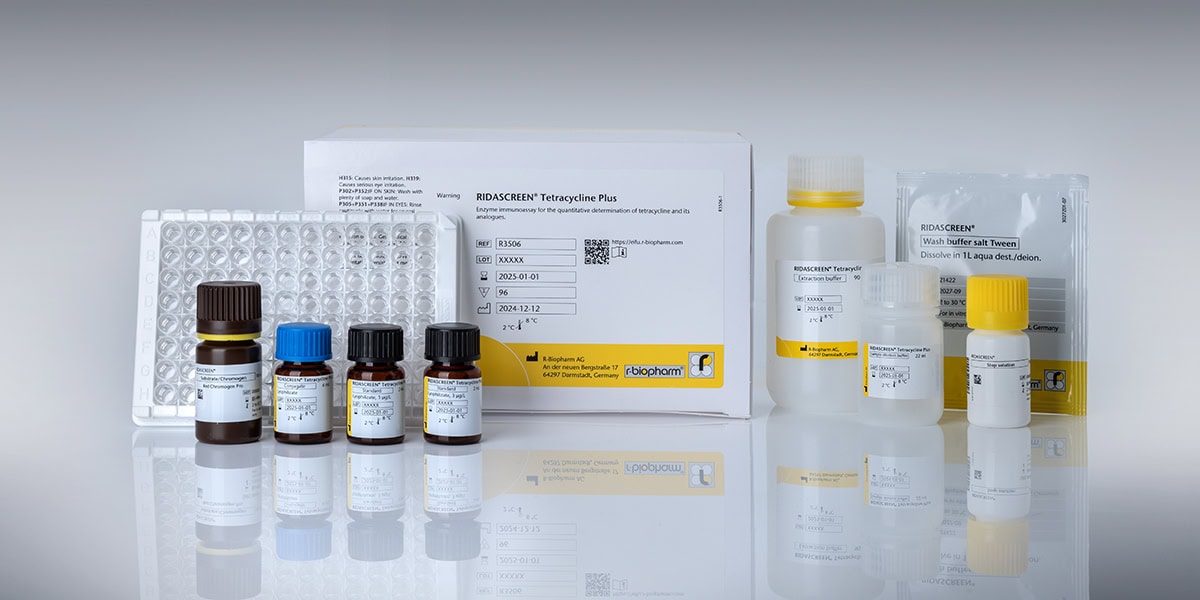
R3506: RIDASCREEN® Tetracyclin
- Simple sample preparation
- Ready-to-use reagents (standards as concentrates)
- Detection limit of approx. 0.8 μg/kg